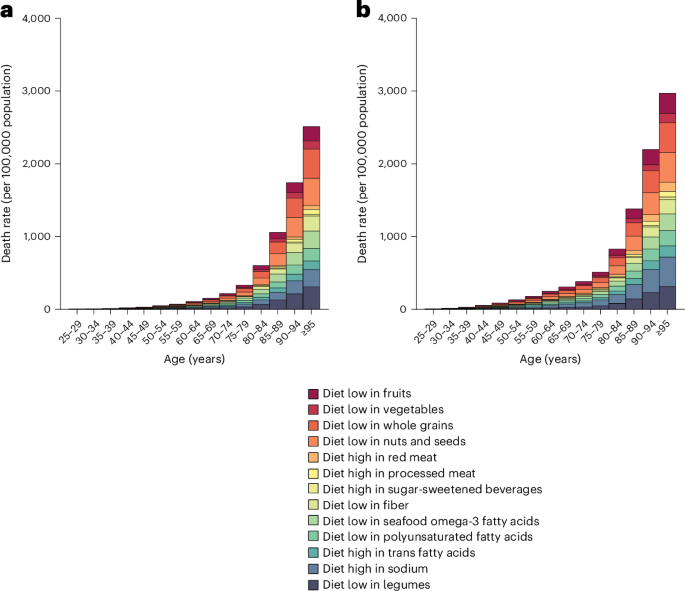
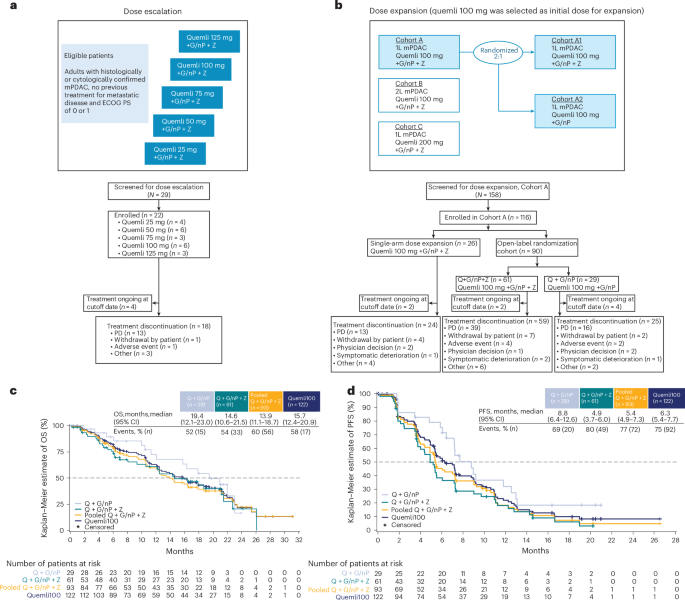
Reliable health information is a fundamental right, Oxford study says
Key Takeaway:
Equitable access to accurate health information is essential for improving global health outcomes and should be a key focus of public health efforts.
What this means for you
This research highlights the importance of access to quality health information. It's early research, so don't change your care yet. Always discuss any health information with your doctor for personalized advice.
Citation:
Nature Medicine - AI Section, 2026. Read article →