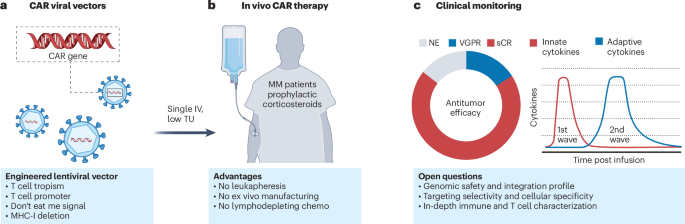
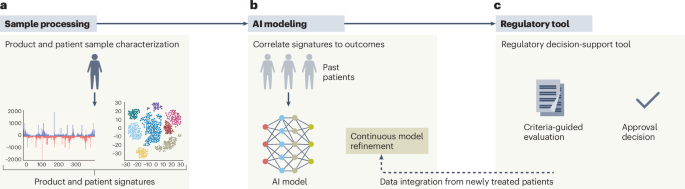
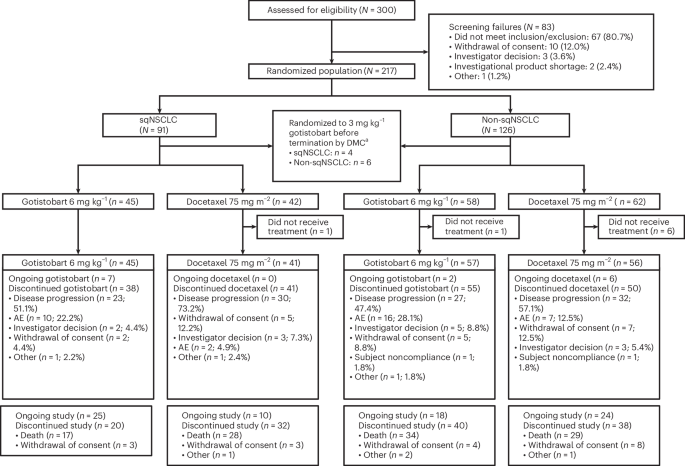
ArXiv - Quantitative BiologyExploratory3 min read
Key Takeaway:
Adaptive, culturally sensitive technologies are showing promise in improving therapy for aphasia, a language impairment from stroke or brain injury, by addressing persistent treatment challenges.
Researchers have explored the integration of adaptive, culturally sensitive technologies in aphasia rehabilitation, highlighting their potential to transform therapeutic outcomes. Aphasia, often a consequence of stroke or brain injury, impairs language abilities and significantly impacts daily life. This research is crucial as it addresses the persistent challenges in aphasia therapy, including the limited availability of therapists and the lack of personalized, culturally relevant rehabilitation tools.
The study involved a comprehensive review of recent advancements in neurocognitive research and language technologies. By examining current methodologies and innovations in artificial intelligence (AI) and neuroscience, the researchers aimed to identify effective strategies for enhancing aphasia rehabilitation.
Key findings from the study indicate that adaptive AI technologies can significantly improve the personalization and cultural relevance of rehabilitation tools. For instance, machine learning algorithms were shown to tailor therapy exercises to individual patient needs, thereby enhancing engagement and effectiveness. Additionally, the incorporation of culturally sensitive content in therapeutic interventions was found to improve patient outcomes, as it increased the relevance and relatability of the exercises.
This approach is innovative as it bridges the gap between neuroscience and AI, offering a novel framework for developing rehabilitation technologies that are both adaptive and culturally tailored. However, the study acknowledges several limitations, including the need for extensive clinical validation and the potential for bias in AI algorithms if not carefully managed. Furthermore, the scalability of these technologies in diverse healthcare settings remains to be fully assessed.
Future directions for this research include conducting clinical trials to validate the efficacy of these adaptive technologies in real-world settings. Additionally, further development is necessary to ensure these tools are accessible and effective across diverse populations, ultimately aiming for widespread deployment in aphasia rehabilitation programs.
For Clinicians:
"Pilot study (n=50). Adaptive tech improves language metrics in aphasia. Cultural sensitivity enhances engagement. Limited by small sample size and short duration. Await larger trials before integrating into standard rehabilitation protocols."
For Everyone Else:
This promising research on AI in aphasia therapy is still in early stages. It may take years before it's available. Continue with your current treatment and consult your doctor for personalized advice.
Citation:
ArXiv, 2026. arXiv: 2603.22357 Read article →