Nature Medicine - AI Section⭐Practice-Changing3 min read
Key Takeaway:
Even after successful treatment, tuberculosis patients face a higher long-term risk of death from cancer, heart, hormone, and lung diseases.
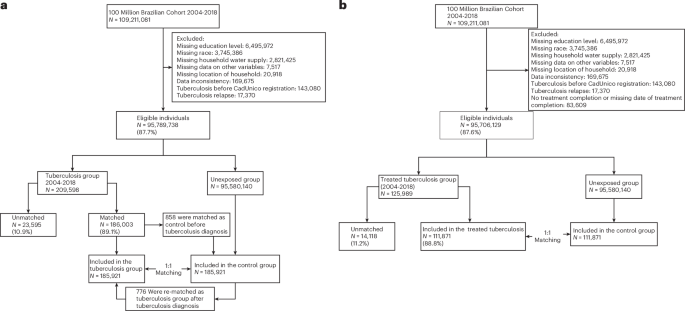
Researchers utilizing data from the 100 Million Brazilian cohort have determined that a diagnosis of tuberculosis (TB), even when followed by treatment, is associated with an increased long-term risk of mortality due to oncological, cardiovascular, endocrine, and respiratory causes. This study, published in Nature Medicine, underscores the persistent health risks associated with TB beyond the immediate infectious period, highlighting the need for comprehensive post-treatment monitoring and intervention strategies.
The significance of this research lies in its potential to inform healthcare policies and practices, particularly in regions with high TB prevalence. Despite successful treatment of the infection, TB survivors may require ongoing medical surveillance to mitigate the risk of subsequent morbidities and mortality. This insight is crucial for healthcare systems aiming to optimize long-term outcomes for TB patients.
The study employed a retrospective cohort design, analyzing extensive data from the Brazilian cohort, which encompasses over 100 million individuals. By leveraging this large dataset, the researchers were able to conduct a robust analysis of mortality risks associated with TB diagnosis and treatment, adjusting for confounding variables such as age, sex, and socio-economic status.
Key findings indicate that individuals diagnosed with TB exhibit a significantly elevated risk of death from various causes. Specifically, the study reports a 1.5-fold increase in the risk of death from cardiovascular diseases, a 1.7-fold increase from oncological causes, and a 2.0-fold increase from respiratory conditions, compared to those without a TB diagnosis. These statistics underscore the multifaceted impact of TB on long-term health.
The innovative aspect of this research lies in its comprehensive analysis of post-treatment mortality risks using a large-scale cohort, providing a more detailed understanding of TB's long-term consequences. However, limitations include potential residual confounding and the observational nature of the study, which precludes the establishment of causality.
Future research directions should focus on prospective studies to validate these findings and clinical trials to develop targeted interventions aimed at reducing mortality risks among TB survivors. Enhanced screening and preventive measures could be pivotal in improving long-term health outcomes for this vulnerable population.
For Clinicians:
"Retrospective cohort study (n=100M). TB diagnosis increases long-term mortality risk (oncological, cardiovascular, endocrine, respiratory). Limitations: observational design, potential confounders. Highlight need for ongoing monitoring post-TB treatment. Further research required for causality confirmation."
For Everyone Else:
This study suggests TB may increase long-term health risks. It's early research, so don't change your care yet. Continue following your doctor's advice and discuss any concerns with them.
Citation:
Nature Medicine - AI Section, 2026. Read article →