Nature Medicine - AI Section⭐Exploratory3 min read
Key Takeaway:
Bispecific T cell engagers, like blinatumomab and teclistamab, show promise in improving symptoms for patients with hard-to-treat autoimmune connective tissue diseases, with good tolerance observed.
Researchers have investigated the use of bispecific T cell engagers, specifically blinatumomab and teclistamab, in a case series involving patients with treatment-refractory autoimmune connective tissue diseases, namely antisynthetase syndrome and systemic sclerosis. The study found that these agents improved disease activity and were well tolerated by patients.
This research is significant as it addresses the therapeutic challenges posed by treatment-refractory autoimmune connective tissue diseases, which often result in poor patient outcomes and limited treatment options. Autoimmune connective tissue diseases, such as antisynthetase syndrome and systemic sclerosis, are characterized by chronic inflammation and progressive tissue damage, necessitating novel therapeutic approaches to improve patient quality of life and disease prognosis.
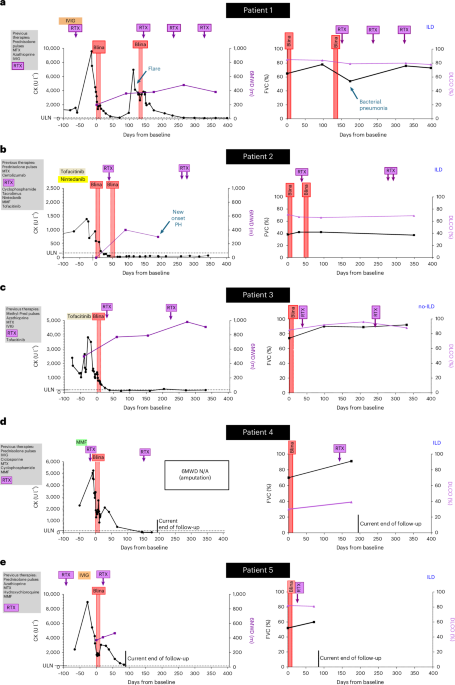
The study was conducted as a case series involving ten patients, five diagnosed with antisynthetase syndrome and five with systemic sclerosis, all of whom were refractory to standard treatments. The patients received bispecific T cell engagers, blinatumomab and teclistamab, which are designed to redirect T cells to target and eliminate pathogenic cells contributing to disease activity.
Results indicated a notable improvement in disease activity as measured by established clinical indices. For instance, patients with antisynthetase syndrome demonstrated a reduction in muscle enzyme levels, while those with systemic sclerosis showed improved skin scores. The agents were well tolerated, with adverse effects being mild to moderate and manageable, thus highlighting their potential as a viable treatment option for these conditions.
The innovation of this approach lies in the application of bispecific T cell engagers, traditionally used in oncology, to autoimmune diseases, representing a novel therapeutic strategy. However, the study is limited by its small sample size and lack of a control group, which restricts the generalizability of the findings and necessitates cautious interpretation.
Future directions should focus on larger, randomized controlled trials to validate these findings and further explore the efficacy and safety of bispecific T cell engagers in a broader autoimmune disease population. This could potentially lead to the development of new therapeutic protocols for treatment-refractory autoimmune connective tissue diseases.
For Clinicians:
"Case series (n=5). Bispecific T cell engagers (blinatumomab, teclistamab) improved refractory autoimmune connective tissue disease activity. Well tolerated. Small sample limits generalizability. Consider cautiously in refractory cases; further research needed for broader application."
For Everyone Else:
Promising early research suggests new treatments might help certain autoimmune diseases. However, these are not yet available. Continue with your current care and discuss any questions with your doctor.
Citation:
Nature Medicine - AI Section, 2026. DOI: s41591-026-04238-4 Read article →