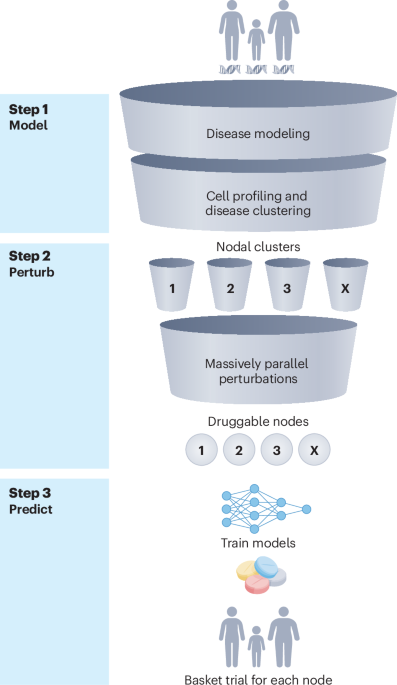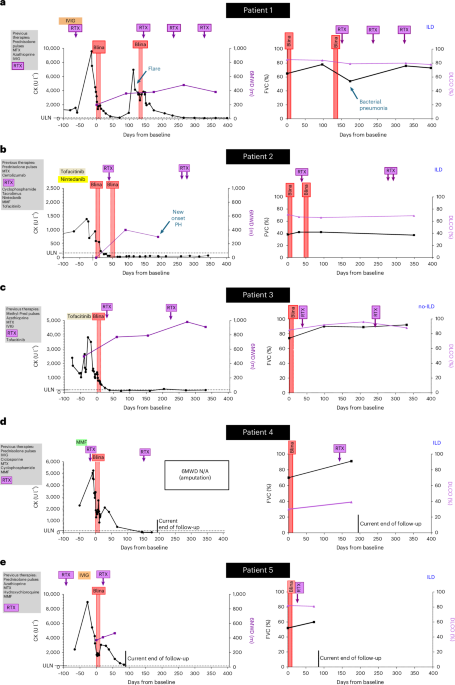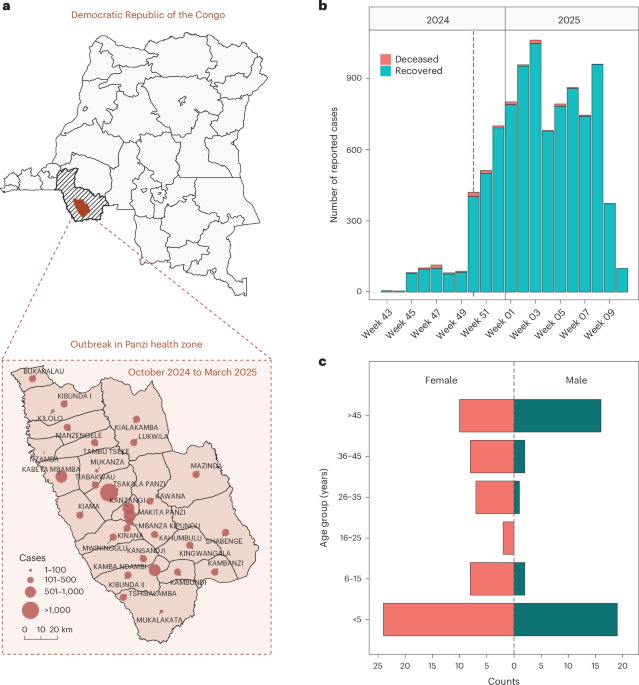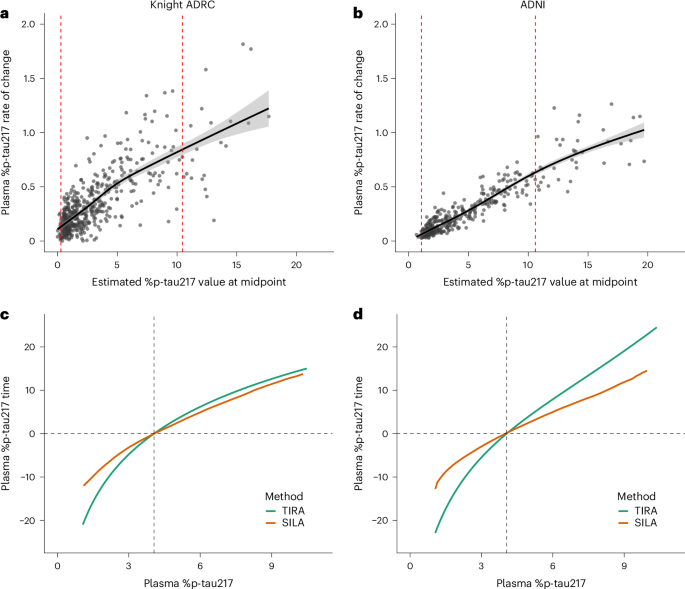
New blood test predicts Alzheimer's symptoms before they start
Key Takeaway:
New blood test using p-tau217 can predict Alzheimer's symptoms in healthy individuals, offering a promising tool for early diagnosis and intervention.
What this means for you
This promising research is still in early stages and not available in clinics. It may take years before it's ready. Continue following your doctor's advice and current care plan for Alzheimer's prevention and management.
Citation:
Nature Medicine - AI Section, 2026. Read article →