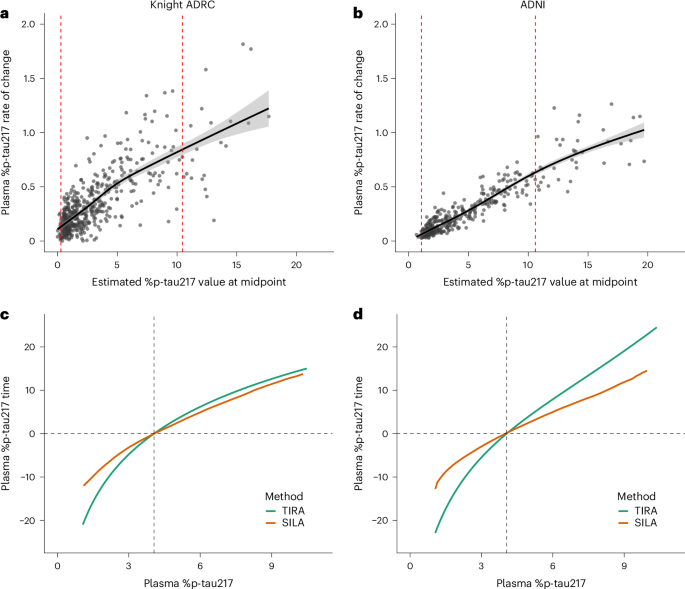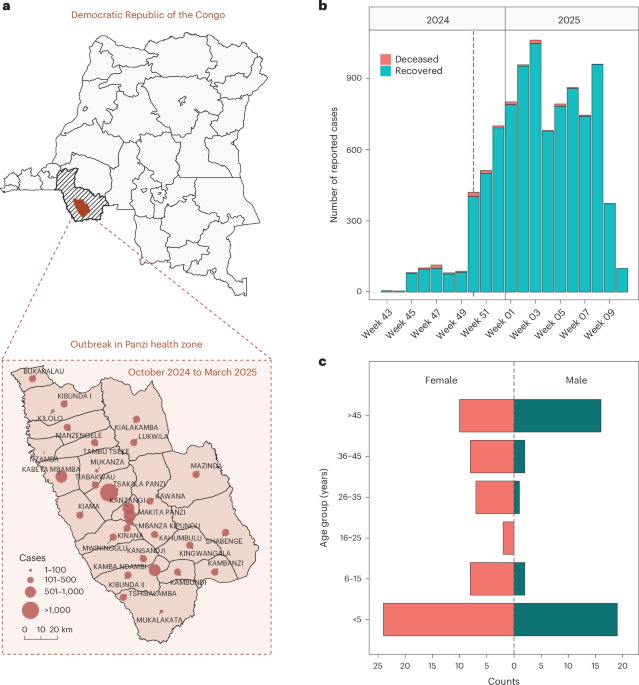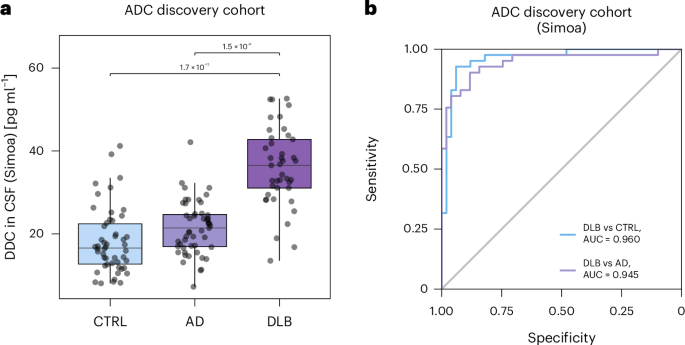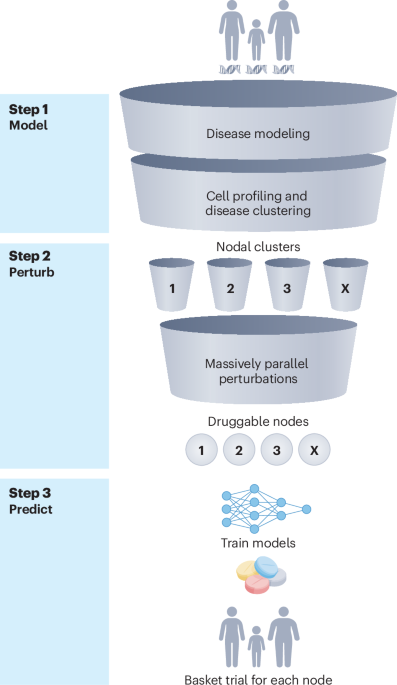
AI finds common treatment targets for rare genetic diseases
Key Takeaway:
AI technology identifies common treatment targets for different genetic diseases, potentially speeding up new drug development for these conditions.
What this means for you
"Exciting early research may lead to new treatments for genetic diseases. However, it's still years away from being available. Please continue with your current care and consult your doctor for guidance."
Citation:
Nature Medicine - AI Section, 2026. Read article →