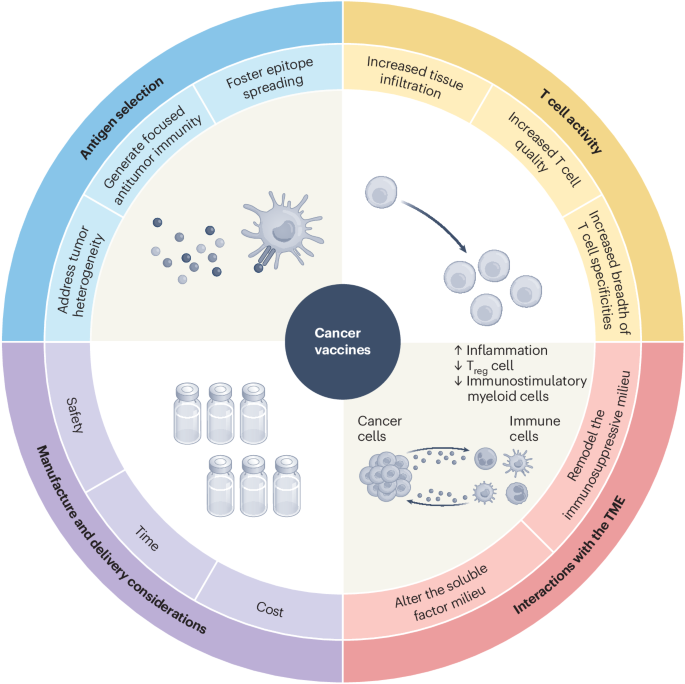
Cracking the code for personalized cancer vaccines
Key Takeaway:
Optimizing cancer vaccines involves selecting the right tumor markers and timing treatments early, which could improve patient outcomes in ongoing clinical trials.
What this means for you
"Exciting early research on cancer vaccines, but it's not yet available for patient care. It may take years to develop. Continue with your current treatment plan and discuss any questions with your doctor."
Citation:
Nature Medicine - AI Section, 2026. DOI: s41591-026-04241-9 Read article →





